Location: Home >> Detail
TOTAL VIEWS
J Sustain Res. 2026;8(2):e260041. https://doi.org/10.20900/jsr20260041
1 Department of Plant and Environmental Sciences, Agricultural Science Center at Farmington, New Mexico State University, 300 Road 4063, Farmington, NM 87401, USA
2 Metropolitan Agricultural Research Center, Colorado State University Spur, Denver, CO 80216, USA
3 Department of Horticulture and Landscape Architecture, Colorado State University, Fort Collins, CO 80523, USA
4 Department of Agricultural Economics & Agricultural Business, New Mexico State University, Las Cruces, NM 88003, USA
* Correspondence: Maria Y. Chavez
Black soldier fly (Hermetia illucens) larvae (BSFL) are popular decomposers capable of reducing food waste and producing frass, a promising substrate additive and fertility product. Frass has gained interest in horticultural production as a more sustainable, partial replacement for fertilizers, and especially peat moss based potting mixes. This has been studied in vegetable and food crops, but has not been thoroughly explored in ornamental crops. Marigolds (Tagetes erecta var. ‘Inca II’) were grown in a peat-moss–based potting mix amended with BSFL frass at 10, 20, or 40% by volume, and were compared to marigolds grown in an unamended peat-based potting-mix control. The BSFL 40% treatment produced negative results for plant size and weight, and replicates in this treatment did not flower. The marigolds grown in the BSFL 10% were statistically comparable to marigolds grown in the control in all parameters. There were no significant differences in relative flavonoid and anthocyanin concentrations of the flowers based on treatment. Electrical conductivity (EC) was significantly higher in the high concentration of frass, BSFL 40%, likely due to the higher sodium concentrations in the frass. The water holding capacity (WHC) of these treatments was also evaluated and results indicate there were no significant differences in the WHC of the treatment percentages. Similar to vegetable studies, low concentrations of frass (10–20%) appear to be an effective media replacement. In order to incorporate larger quantities of frass into crop production, alternative approaches to frass use must be considered. Washing frass to reduce sodium concentrations or altering larval diet to reduce ammonium concentrations should be considered. This project warrants more research in pretreating frass in horticultural production prior to application.
BSFL, black soldier fly larvae; CEA, controlled environment agriculture; DW, dry weight; SC, stomatal conductance; PSH, plant size at harvest; EC, electrical conductivity; RCC, relative chlorophyll content; WHC, water holding capacity
Black soldier fly (Hermetia illucens) larvae (BSFL) are highly efficient decomposers [1]. Insect producers use side streams from other industries, such as restaurants and brewery waste, as feed for the larvae, which is converted into a high protein product during larval digestion [2]. By reusing food sources and recycling nutrients to create a high protein feed for other livestock, producers are reducing food costs and closing gaps in a circular economy [3]. Additionally, during the mass industrial rearing of insects, frass is produced in large quantities. Rather than allowing the frass to accumulate as waste, there is a growing interest in recycling these nutrients as horticultural amendments [4]. This has positive economic and environmental benefits for farmers and producers [5] and improves nutrient redirection in the food system [3].
BSFL frass has been studied in controlled environment agriculture (CEA) crops such as tomatoes [6,7], peppers [8], leafy greens [7,9–11], and sweet potatoes [12,13]. In most vegetable studies, low rates of frass incorporation have positive or neutral impacts on crop production and quality, while high rates of incorporation can produce negative outcomes for crop production. The seedling emergence of tomatoes, lettuce, and arugula has also been evaluated with frass treatments, indicating that frass in low concentrations can be effective as an amendment in starting media [14]. CEA includes the production of plants in structures such as high tunnels, greenhouses, growth chambers, or warehouses for indoor vertical farming [15]. CEA provides an alternative approach for food production with protection from natural environmental factors such as pests, disease, and extreme weather events. Greenhouses are also popular in urban areas with less land for crop production [16]. Recently, the number of US greenhouse producers has increased by 71%, and these numbers are expected to increase in the future [17].
Ornamental plants are commonly grown in CEA using soilless substrates often comprised of a high percentage of peat. Peat is an ideal substrate for crop growth due to its water holding capacity (WHC) and loamy texture and is common in greenhouse and gardening operations [15]. Additionally, as popularity of CEA increases, the use of peat-based substrates is increasing as well, and peatlands have been decreasing by 0.05% globally each year [18]. Peat is found in marsh environments, including decaying organic and inorganic components [19,20], and unfortunately, peat mining can result in biodiversity loss and greenhouse gas emissions [21,22]. Mined peat marshes experience disturbances to their methanotrophic bacteria, which are capable of filtering methane. When that filter is broken, peat marshes begin to emit methane into the atmosphere, increasing air pollution and greenhouse gas emission [23]. Wildlife diversity and richness also see significant reductions after peat mining occurs [24]. Furthermore, peat moss has also been increasing in price [25]. Therefore, alternatives to peat are a growing interest for ornamental producers [26].
In an effort to reduce reliance on peat for ornamental production, a few studies have investigated frass as a fertility amendment and alternative substrate. Mealworm (Tenebrio molitor) frass (MWF) and hen (Gallus gallus domesticus) manure were mixed into a growing mix to form two separate treatments for nasturtiums (Tropaeolum majus), zinnias (Zinnia elegans), and dwarf sunflowers (Helianthus gracilentus) and were compared to a control without MWF or hen manure [27]. Flowers per unit of all varieties, height and flower diameter of the dwarf sunflower, and overall biomass of nasturtium were significantly higher in the hen manure and MWF treatments compared to the control. Since manure is a common organic fertilizer, the lack of statistical significance indicates that insect frass can produce comparable results to an already adopted practice. The presence of some detritivores, like BSFL, can also reduce volatile organic compounds [28] and pest presence [29] in waste streams. Therefore, this is promising for the inclusion of frass in organic operations.
A study by Beasley et al. (2023) [30] utilized a conventional fertilizer control to provide more insight into the ability of frass to replace inorganic, chemical fertilizers in ornamental production. Coleus (Plectranthus scutellarioides) plants were grown in containers with increasing rates (0, 0.1, 0.2, and 0.3 kg/m3 N) of standard fertilizer (SF) and BSFL frass fertilizer. Shoot dry weight was significantly higher in the 0.3 SF treatment compared to the 0.3 BSFL. At all other rates, SF and BSFL were statistically comparable. However, cumulative nitrogen leaching was significantly lower in the BSFL treatment compared to the SF. This indicates that an organic side stream, such as frass, can not only match synthetic fertilizers performance, but may also reduce negative environmental consequences, such as nutrient runoff. Other studies have utilized insect frass in evaluating flowering plants, herbivory, and pest survival [31,32]. Results from these studies indicate that flower number increases in black mustard, regardless of herbivore activity [31], and field mustard pest populations decreased with BSFL frass treatments [32].
Marigolds (Tagetes) are a genus of angiosperms native to North and Central America. While they are aesthetically and olfactorily pleasing for garden and landscaping design, they also have a large variety of cultural, medicinal, anti-pathogenic, and food related uses [33]. To our knowledge, insect frass has not been thoroughly investigated for marigold production; however, other studies have observed alternative peat replacements in marigold production. For example, vermicompost was incorporated into a potting medium for African Marigold (Tagetes erecta) at 10, 20, 30, 40, 50 and 60% [34]. The 60% treatment produced the highest fresh and dry weight of the shoots and roots, while also producing the greatest number of florets and photosynthetic pigments.
The objective of this study was to explore the impact of insect frass as a partial replacement of peat based potting mixes on the floral and plant growth characteristics of African marigolds (Tagetes erecta var. ‘Inca II’ Hybrid Color Mix) (Figure 1). Additionally, we wanted to examine the physical components, water holding capacity (WHC) and porosity, of insect frass when amended into peat based potting mixes. We hypothesize that growth and flowering parameters of marigolds grown in BSFL will be comparable to those grown in a 100% potting mix control. We also hypothesize that frass-based treatments will have higher or equal WHC and porosity compared to a 100% peat-based potting mix control.
The experiment took place in Denver, CO, USA at the Colorado State University Spur Terra greenhouse, elevation 1684.02 m (en-us.topographic-map.com). Greenhouse temperature and relative humidity set points were 17–25 °C and 40–75%, respectively. African marigold ‘Inca II’ (Park Seed Wholesale, Greenwood, SC, USA) seedlings were sown on September 21, 2023 in 84-cell trays in a peat-vermiculite germination mix (Lambert LM-GPS, Rivière-Ouelle, QC, Canada). They were watered once to twice a day, as needed. Seedlings were transplanted into 500 mL pots on October 27, 2023, in treatment mixtures consisting of a combination of peat mix [Berger BM6 peat blend (Saint-Modeste, QC, Canada)] and BSFL frass, reared mostly on spent brewery grains (EVO Conversion Systems, College Station, TX, USA) (Table 1).
The experiment was a completely randomized design (CRD) using four levels total, three treatments of each frass amended potting mixture and a 100% peat control (Table 2), with eight replicates per treatment.
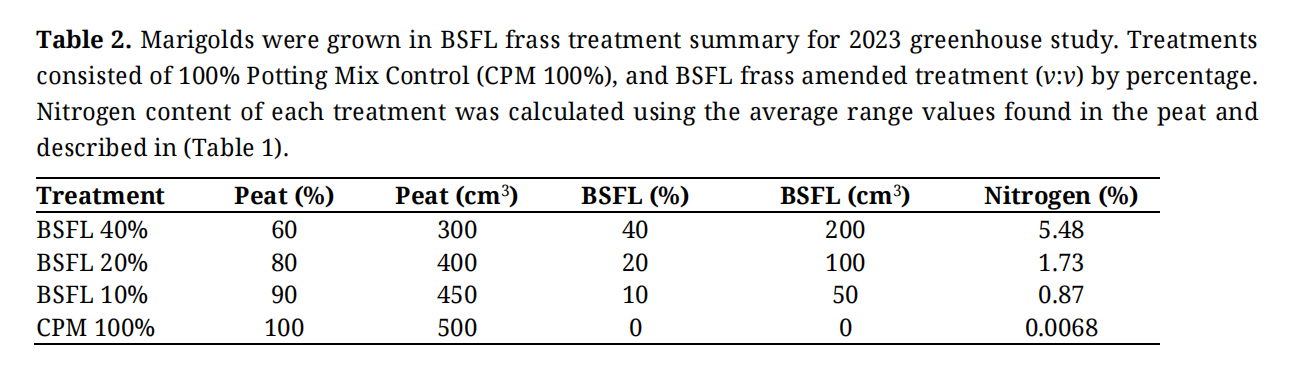 Table 2. Marigolds were grown in BSFL frass treatment summary for 2023 greenhouse study. Treatments consisted of 100% Potting Mix Control (CPM 100%), and BSFL frass amended treatment (v:v) by percentage. Nitrogen content of each treatment was calculated using the average range values found in the peat and described in (Table 1).
Table 2. Marigolds were grown in BSFL frass treatment summary for 2023 greenhouse study. Treatments consisted of 100% Potting Mix Control (CPM 100%), and BSFL frass amended treatment (v:v) by percentage. Nitrogen content of each treatment was calculated using the average range values found in the peat and described in (Table 1).
All treatments were watered once to twice a day and fertigated weekly with Tower Farms Dry Mineral Blend Fertilizer (Tower Garden LLC, Collierville, TN, USA), chemical composition (Table 1). Data collection for all treatments occurred on December 19, 2023. Plant size at harvest (PSH) was calculated by measuring plant width [the greatest width (W1) and the perpendicular width (W2); cm] and height (cm) using Equation (1) [10]:
Stomatal conductance (SC) was measured with a SC-1 Leaf Porometer System (Meter Group, Pullman, WA, USA) and relative chlorophyll concentrations (RCC) were taken with a handheld meter (atLEAF CHLBLUE, Wilmington, DE, USA) before harvest. atLeaf values were converted to SPAD and then mg/cm2 using the conversion tool on the atLeaf website (https://www.atleaf.com/SPAD). Fully opened flowers and non-bloomed flowers, or florets, were counted to record floret number. Total relative flavonoid and anthocyanin concentrations were measured from fully opened flowers with an MPM-100 multipigment reader (Opti-Sciences, Hudson, NM, USA). Fully opened flowers were separated from the rest of the plant, placed in paper bags, and dried in a Heratherm drying oven (Thermo Fisher Scientific, Waltham, MA, USA) separately from the rest of the plant at 60OC Both flower and plant dry matter were weighed after 72 h in the drying oven (Mettler Toledo, Denver, CO, USA).
Post destructive harvest, electrical conductivity (EC) and pH measurements were taken using the pour through method [35] on December 22, 2023. The pots were irrigated as usual and drained for a minimum of 30 min. A volume of 100 mL of distilled water was poured into each pot and let to drain in a glass beaker, where the leachate was collected. The leachate was then measured with a pH/EC combo meter (Bluelab Corporation, Industrial Park, Tauranga, New Zealand).
Further, water holding capacity (WHC) and porosity of the substrate treatments were evaluated on December 1, 2023, separately from the main experiment. This evaluation followed a CRD, similar to Table 2, but included the 100% frass control to fully evaluate the design, producing five levels total and four replicates per treatment. Treatments were mixed, placed in paper bags, and dried in a drying oven for 72 h at 70 °C. On December 4, 2023, dried substrates were weighed and placed in 500 mL pots. 150 mL of distilled water was slowly poured through each treatment and drained into plastic beakers and allowed to drip for approximately two minutes. Pour through liquid was measured and the wet substrates were left to sit in 3.8 L buckets. On December 6, 2023, 48 h later, saturated treatments were weighed. WHC and porosity were calculated with Equations (2) and (3), respectively:
Statistical analysis was conducted using JMP®, Pro 17 (SAS Institute Inc., Cary, NC, 1989–2023, USA) and R Studio (Boston, MA, USA) 2025.05.1 (R version 4.4.2). To evaluate water holding capacity and porosity, percentages were converted into proportions and transformed with a logit transformation. The Anderson-Darling test for normality and Levene’s test for equal variance were conducted to determine if the assumptions for an ANOVA were met. A one-way ANOVA was used to analyze the differences between treatments and were followed by a post hoc Tukey’s HD multiple comparison test when appropriate. A threshold of p < 0.05 was significant.
Because differences in flower color among plants were not evident at the beginning of the experiment, a flower color metric was incorporated into the analysis to account for these differences as flower development occurred. A color value was assigned to each plant to quantify variation in yellow intensity. This color metric was included as a covariate to explain a portion of the variability among plants pertaining to flower color and to isolate, as much as possible, the effects of the treatments.
The analysis of covariance was performed in SAS 9.4 using proc GLIMMIX for the following variables: PHS, PDW, EC, pH, SC, SPAD, FDW, FLORET (under the assumption that this response follows the Poisson distribution), FLAV, and ANTH. Mean separation was conducted through the postprocessing procedure PLM, with Tukey’s method used to control for multiplicity at the 5% significance level. Depending on the Bayesian Information Criterion (BIC) metric, that evaluates model fit, the covariate effect was either made treatment specific or only entered the model as a main effect.
Plant size at harvest (Figure 2A) and plant dry weight (Figure 2B) were greatest in the control and BSFL 10% treatment and lowest in the BSFL 40% treatment (F = 50.30; p < 0.0001, F = 55.56; p < 0.0001). Relative chlorophyll concentration (Figure 2C) was significantly higher in the BSFL 10% and lowest in the BSFL 40% (F = 4.20; p = 0.01). Stomatal conductance (Figure 2D) in the BSFL 20% treatment was significantly higher than the control and BSFL 40% (F = 8.50; p = 0.0004).
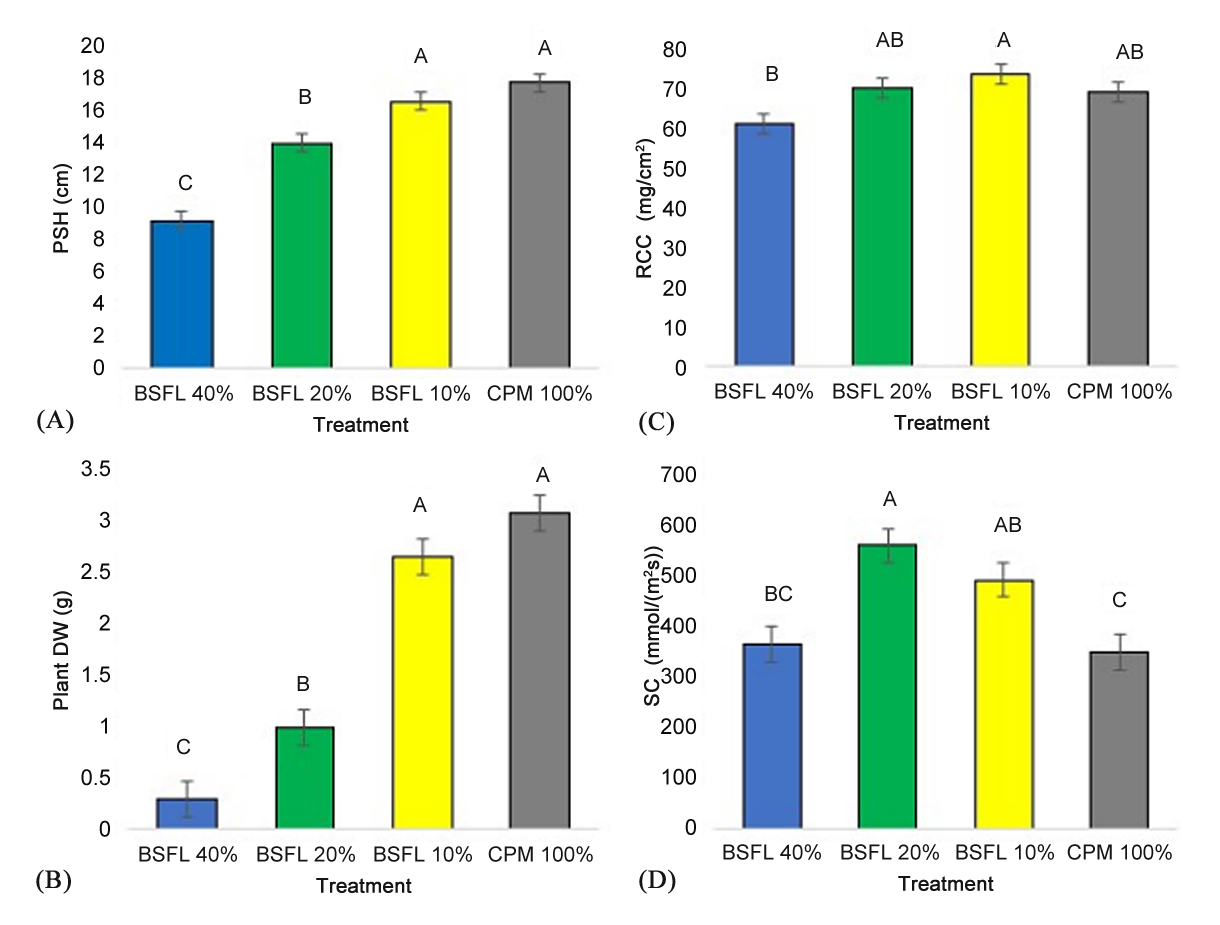 Figure 2. Plant Size at Harvest (PSH) (A), Plant Dry Weight (DW) (B), Relative Chlorophyll Concentrations (RCC) (C), and Stomatal Conductance (SC) (D) ±SEM of marigolds grown in BSFL frass. Marigolds were grown in a 100% potting mix control (CPM 100%: grey column) and three treatments with BSFL frass in partial peat replacements (BSFL 10% treatment: yellow columns, BSFL 20%: green columns, and BSFL 40%: blue columns). Different letters (A–C) indicate significant differences between treatments (α = 0.05), ANOVA followed by Tukey’s HSD.
Figure 2. Plant Size at Harvest (PSH) (A), Plant Dry Weight (DW) (B), Relative Chlorophyll Concentrations (RCC) (C), and Stomatal Conductance (SC) (D) ±SEM of marigolds grown in BSFL frass. Marigolds were grown in a 100% potting mix control (CPM 100%: grey column) and three treatments with BSFL frass in partial peat replacements (BSFL 10% treatment: yellow columns, BSFL 20%: green columns, and BSFL 40%: blue columns). Different letters (A–C) indicate significant differences between treatments (α = 0.05), ANOVA followed by Tukey’s HSD.
The flower response variables were only evaluated for treatments where flowering occurred: CPM 100%, BSFL 10%, and BSFL 20%. Dry weight of the marigold flower (Figure 3A) and floret number (Figure 3B) was greater in the BSFL 10% treatment and control compared to the BSFL 20% treatment (F = 8.09; p = 0.0025, F = 12.42; p ≤ 0.0001). Differences in relative flavonoid (F = 1.22; p = 0.31) and anthocyanin (F = 1.51; p = 0.24) concentrations (Figures 3C,D) were not statistically significant between treatments and/or control.
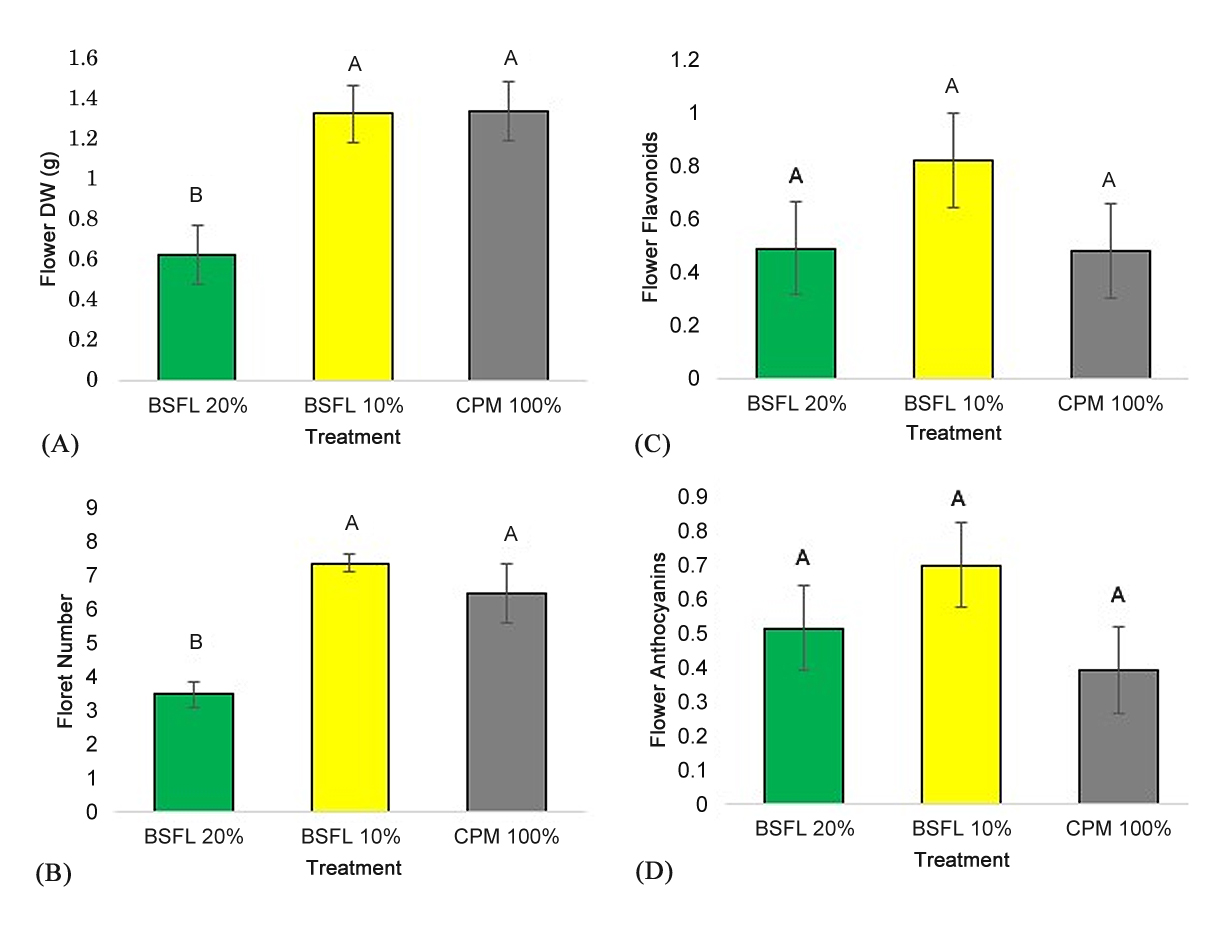 Figure 3. Flower Dry Weight (DW) (A), Floret Number (B), and Relative Flavonoid (C) and Anthocyanin Concentrations (D) ±SEM of marigolds grown in BSFL frass. Marigolds were grown in a 100% potting mix control (CPM 100%: grey column) and three treatments with BSFL frass in partial peat replacements (BSFL 10% treatment: yellow columns and BSFL 20%: green columns). Different letters (A–B) indicate significant differences between treatments (α = 0.05), ANOVA followed by Tukey’s HSD.
Figure 3. Flower Dry Weight (DW) (A), Floret Number (B), and Relative Flavonoid (C) and Anthocyanin Concentrations (D) ±SEM of marigolds grown in BSFL frass. Marigolds were grown in a 100% potting mix control (CPM 100%: grey column) and three treatments with BSFL frass in partial peat replacements (BSFL 10% treatment: yellow columns and BSFL 20%: green columns). Different letters (A–B) indicate significant differences between treatments (α = 0.05), ANOVA followed by Tukey’s HSD.
The African Marigold variety ‘Inca II’ was selected for this experiment. While the variety is stated to be uniform in height, habit, and bloom size, the seed utilized included a mix of flower colors. While seed mixes such as this are commonly used in commercial production, flower color can be an indicator of additional variation in plant phenotype. To ensure that flower color was the only trait showing inherent variation in the present experiment, an additional analysis of flower color was conducted for each parameter. Treatments resulting in flower development were evaluated, with flower color as a covariate in an ANCOVA analysis (Table 3). The importance of the interaction term was evaluated based on a model-fit criterion (BIC) which determined whether to retain or remove it. Flower color was only significant in the floral relative anthocyanin concentrations (Table 3), whereby an increase in anthocyanin concentrations was observed for orange flowers compared to yellow flowers, but gold flowers were statistically comparable to both yellow and orange flowers. For the parameters where flower color and treatment interaction were analyzed, there were no significant interaction effects. Of note is the effects of treatment were not of interest for further discussion as this analysis only included the treatments where flowering occurred, and not the BSFL 40%.
EC (Figure 4A) after the study was completed was highest in the BSFL 40% leachate, then BSFL 20%, and lowest in the BSFL 10% and control (F = 51.98; p ≤ 0.0001). The opposite trend occurred for pH (Figure 4B), where BSFL 40% was significantly lower than BSFL 10% (F = 3.18; p = 0.04).
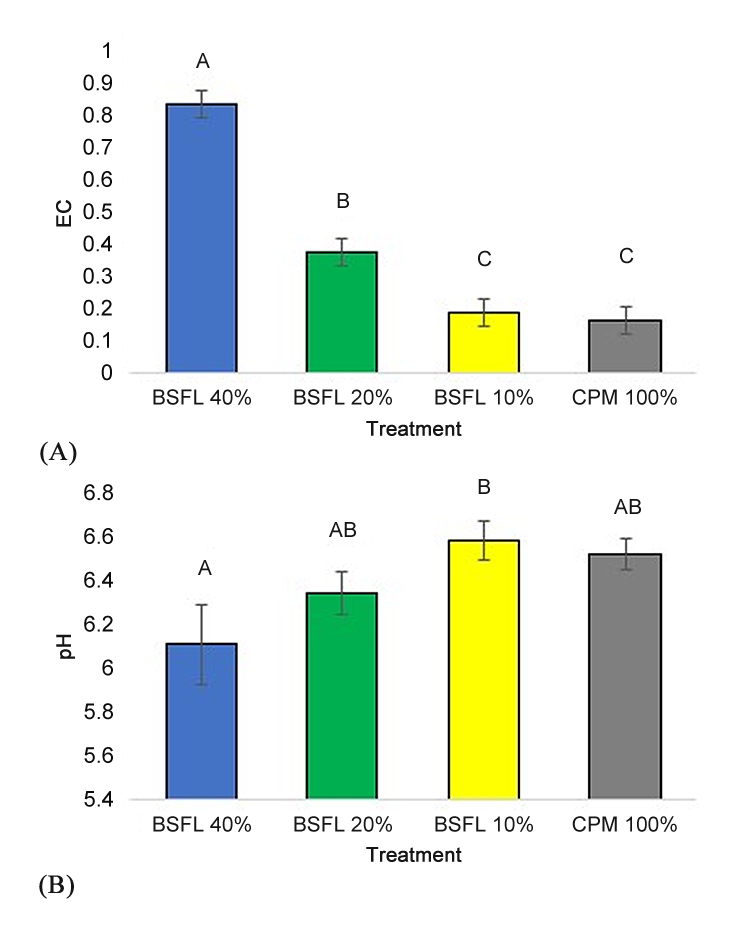 Figure 4. Electrical Conductivity (EC) (A), and pH (B) ± SEM of BSFL frass amended potting mix. Marigolds were grown in a 100% potting mix control (CPM 100%: grey column) and three treatments with BSFL frass in partial peat replacements (BSFL 10% treatment: yellow columns, BSFL 20%: green columns, and BSFL 40%: blue columns). Different letters (A–C) indicate significant differences between treatments (α = 0.05), ANOVA followed by Tukey’s HSD.
Figure 4. Electrical Conductivity (EC) (A), and pH (B) ± SEM of BSFL frass amended potting mix. Marigolds were grown in a 100% potting mix control (CPM 100%: grey column) and three treatments with BSFL frass in partial peat replacements (BSFL 10% treatment: yellow columns, BSFL 20%: green columns, and BSFL 40%: blue columns). Different letters (A–C) indicate significant differences between treatments (α = 0.05), ANOVA followed by Tukey’s HSD.
There were no significant differences in WHC (Figure 5A) or percent porosity (Figure 5B) between treatment means (F = 0.50; p = 0.74, F = 1.44; p = 0.27).
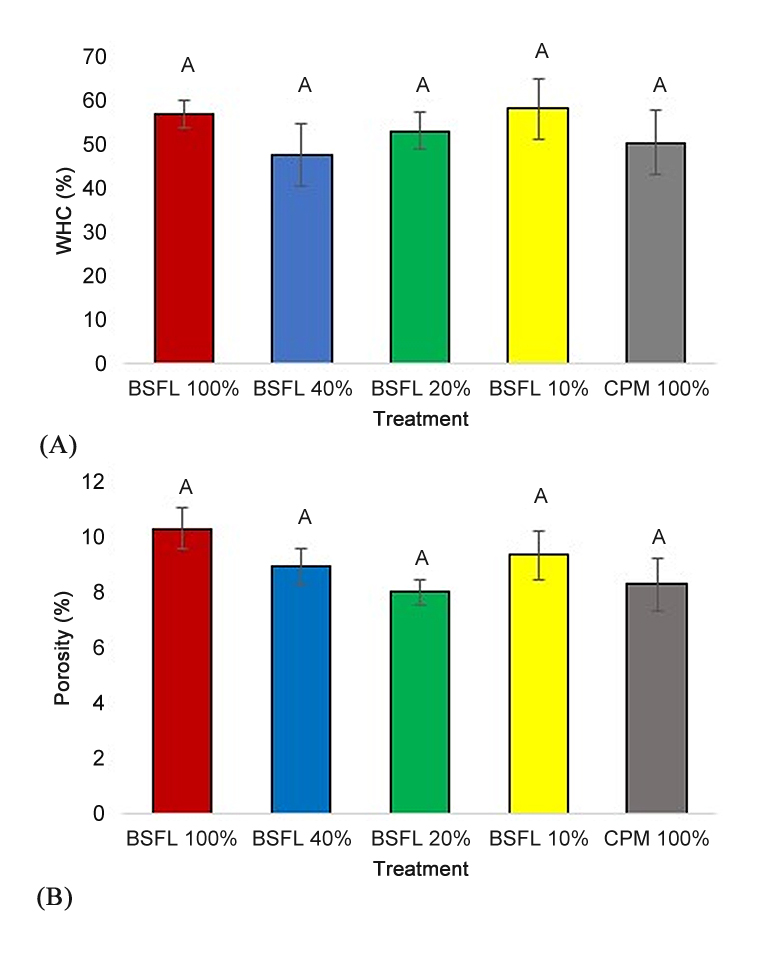 Figure 5. Water holding capacity (WHC) (in percentage) (A) and porosity (B) (in percentage) ± SEM of BSFL frass treatments. WHC was tested in a 100% potting mix control (CPM 100%: grey column), a 100% frass control (BSFL 100%: red column), and three treatments with BSFL frass in partial peat based potting mix replacements (BSFL 10% treatment: yellow columns, BSFL 20%: green columns, and BSFL 40%: blue columns). Letter (A) indicates no significant differences between treatments and controls (α = 0.05).
Figure 5. Water holding capacity (WHC) (in percentage) (A) and porosity (B) (in percentage) ± SEM of BSFL frass treatments. WHC was tested in a 100% potting mix control (CPM 100%: grey column), a 100% frass control (BSFL 100%: red column), and three treatments with BSFL frass in partial peat based potting mix replacements (BSFL 10% treatment: yellow columns, BSFL 20%: green columns, and BSFL 40%: blue columns). Letter (A) indicates no significant differences between treatments and controls (α = 0.05).
The BSFL 10% treatment was comparable to the control in all parameters, except stomatal conductance (SC). This indicates that low amounts of frass incorporation can have positive or neutral results for ornamental growth and production, similar to what has been seen in food crops [6,7,9,10]. Benefit cost analysis of sweet potato slips grown in BSFL frass reduced operation costs by one to two thousand dollars a year [12]. Over time these reductions could possibly accumulate into savings, while also reducing the harmful consequences of peat extraction, resulting in a more circular economy [3] for insect producers and ornamental plant growers.
Reduced PSH observed in the higher frass treatments (BSFL 40%) may have been due to several factors including higher concentrations of soluble salts, ammonium toxicity, or the effects of microbes. For soluble salts, sodium (Na) was observed in high concentrations through the frass analysis (Table 2). Additionally, EC was highest in the BSFL 40% treatment leachate at the end of the study. Increases in Na uptake [9,10] and EC [36] with frass applications have been reported in several vegetable studies and are known to produce harmful outcomes for plant growth, like reduced plant size, weight, and, in high enough concentrations, reduce survival [37]. Additionally, the higher proportion of nitrogen present as ammonium in the frass analysis (Table 1) possibly reduced plant dry weight, as excessive ammonium has been shown to limit marigold growth [38]. Specifically, marigold height, fresh weight and dry weight were found to be highest when nitrate and ammonium were balanced (50/50) in the fertilizer applied. Higher proportions of ammonium reduced marigold dry weight significantly [39]. Increased ammonium concentrations relative to nitrates were observed in frass derived from food waste [40]. This has also been identified as a potential issue in other frass treated studies, correlating with decreased spring onion and sweet corn growth [41]. Additionally, studies indicate that BSFL fed varying diets produced the highest soil nitrate and ammonium in a high protein and N diet [42]. This was also connected to higher CO2, N2O, and NO fluxes in soils, which could lead to an increase in greenhouse gas emissions. Microbially, the high carbon content of frass may lead to high biological oxygen demand resulting in low oxygen conditions in the root zone and potential nitrogen immobilization, which can negatively affect plant growth [39]. Though marigolds are known for their anti-pathogenic compounds and bioremedial properties [43], several microoganisms are known to negatively impact marigold production, such as Cucumber mosaic virus (Cucumovirus CMV) [44] and black spot (Alternaria sp.) [45]. These can be introduced to marigolds through sap [44], wind, water, or direct contact, and can be exacerbated by weather conditions [45]. Additionally, bacterial abundance and diversity may increase in frass treated substrates compared to those treated with conventional urea fertilizers [46], which could have negative or positive outcomes for marigold growth and production. Ultimately, while frass introduction into a greenhouse substrate may alter the plant microbiome, further research isolating individual microorganisms and their interaction with the plant is needed, especially when pathogens have been identified as a concern in the prospective crop selection.
Lack of significant differences pertaining to the physical properties of the treatments, WHC and porosity, further informs the inference that chemical, microbial, or a combination of these factors may be the cause of negative plant outcomes at high percentages of frass inclusion. It is also important to note that the WHC analysis was conducted with fresh media and frass containing no plant material. Thus, it is possible that longer term effects and interactions may occur over the course of crop production in a greenhouse [39]. Interactions with the root system, irrigation, and fertilizer applications will likely impact WHC throughout the duration of both the plant and frass life span. Therefore, future studies should include a WHC analysis conducted throughout the duration of the experiment and at the end. Additionally, SC was lowest in the control and highest in the BSFL 20% treatment. This result differs from the trend observed in the other parameters, but may be due to increased water availability, which generally results in increased water use [47]. The BSFL 40% treatment, highest in EC, did not flower and produced the smallest plants. While the lack of growth observed in this treatment may have led to reduced SC, the high Na observed in the frass may also have led to salt stress resulting in a reduced SC (Table 1). The presence of high salt concentration resulting in stress is further indicated by higher EC observed in the BSFL 40% leachate (Figure 4). Based on PSH and dry weight results, the BSFL 20% treatment also appears to be a frass incorporation rate high enough to induce stress and reduce plant productivity and flowering, albeit not as detrimental as BSFL 40%. However, the BSFL 20% treatment resulted in the highest SC observed in this study. This increase in SC may also be a response to salinity stress, which has been observed in iceberg lettuce (Lactuca sativa var. Capitata)) through a decrease plant water use efficiency [48] as well as an increase in stomatal opening in tomatoes (Solanum lycopersicum var ‘Capello’) [49].
Despite frass having a higher pH than the peat, the BSFL 40% treatment leachate had the lowest pH. This is likely due to the poor root system (and overall growth) of the plants in this treatment and the subsequent decrease in fertilizer uptake, which was primarily composed of nitrates (Table 1). Nitrate uptake by plants increases pH over time as the plants release OH into the substrate. Therefore, actively growing plants should experience an upward trend in pH, whereas poorly developed plants, would produce a lower pH environment in comparison. Ammonium uptake by plants conversely releases H+ into the substrate. Over time, the high ammonium present in the frass is also likely contributing to a reduced pH in high frass treatments [50].
Given frass has the physical attributes, as they pertain to porosity and WHC at the start of the study, to promote plant growth, adjustments can be made to mitigate other issues. Coconut coir has become a common component in many greenhouse substrates. However, due to its high concentration of sodium, the material must be pretreated. Washing coconut coir before incorporation has become an effective and common practice for producers and consumers [51]. A study utilizing BSFL frass amendments in ornamental production included a similar washed treatment to reduce initial sodium concentrations. This treatment reduced peat inclusion by 40% and produced results statistically comparable to the control in plant size, shoot weight, root weight, flower weight, and chlorophyll concentrations [52]. Treatments (chemical, physical, or thermal) prior to frass application may also change microbial interactions in the potting mix. For example, frass is commonly heat treated after digestion to reduce foodborne pathogens, such as Salmonella [53]. This also emphasizes the importance of the larval diet, which has been observed to impact the microbial communities present in frass [54]. Altering larval diet has also been an effective way of reducing ammonium toxicity in frass treated plants [55] and studies with low initial concentrations of ammonium in the frass produced leachate samples that did not contain ammonium [10]. Thus, exploring methods to reduce ammonium post rearing is another area of future research. Pre-treating the frass prior to incorporation could allow for greater percentages to be introduced in potting mixes, further reducing costs and concerning environmental practices.
Quality parameters, such as relative flavonoid and anthocyanin concentrations of the flower petals, were statistically comparable across all treatments and the control. In contrast, kale (Brassica oleracea cv. Blue ridge) grown in a peat-based substrate amended with 60% and 40% BSFL frass produced significantly higher concentrations of flavonoids compared to the 100% peat control [56]. Thus, the impact of BSFL frass on plant quality parameters may be species specific. Flavonoid concentrations in the diet of insects have been seen to increase the flavonoid concentrations in the resulting frass [57], further emphasizing the importance of larval diet management in insect production. Flavonoids are secondary metabolites associated with producing positive health outcomes for humans [58]. Similar studies observed increased anthocyanin concentration in red cabbage (Brassica oleracea) microgreens when plants were fertilized with insect frass [59]. The larvae were reared on a diet of both brewery grains and okra, further supporting that larvae diet may impact crop results from frass incorporation. Anthocyanins are plant antioxidants that can serve a variety of purposes including dyes and medicines [60]. Due to the pigmented nature of the compound, it is reasonable to expect results would vary with the varying marigold flower colors utilized in the present study and that fewer treatment effects would be observed. Since marigolds are often grown for medicinal purposes [33], the lack of differences between treatments and control in relative flavonoid and anthocyanin concentrations is promising for frass utilization, especially at the 10% incorporation rate, since the flower dry weight was also comparable to the control. Increasing antioxidants in food and medicinal crops may be important for future research to increase human health outcomes [61]. This may be especially beneficial if sourced from insect frass, as it may also improve agricultural sustainability and resiliency [62]. Additionally, each plant in this study produced one full flower and several florets, or unbloomed flowers. Floret number was also highest in BSFL 10% treatment, possible providing further incentive for the incorporation of frass into substrate mixes for commercial production.
Marigolds have been a large component of the floriculture industry for decades, especially across Asia [63]. 600,000 tons of marigolds are produced globally each year, approximately 75–80% by India, 80% of which is consumed locally [43]. In India, marigolds and most other crops are grown outdoors. Due to the high initial costs, only 5000 ha is currently operating as CEA in the country [64]. Interestingly, studies have observed significant increase in marigold height, weight, yield, and flower number when grown in a controlled environment compared to a conventional outdoor system [65]. Incorporating frass into CEA substrates presents a valuable opportunity to reduce both peat and synthetic fertilizers. Ultimately, redirecting waste streams from one cycle of production to another (Figure 6) can decrease material sent to the landfill, prevent natural resource extraction, protect vulnerable ecosystems [66], and improve circular economies [3].
 Figure 6. The circular economy relationship for insect producers, crop growers, and consumers. Insect producers use food waste to feed larvae and create a high protein product. Leftover frass can be utilized as a horticultural fertilizer or amendment during crop production. Crop growers sell or donate their products to other food partners, such as restaurants and grocery stores.
Figure 6. The circular economy relationship for insect producers, crop growers, and consumers. Insect producers use food waste to feed larvae and create a high protein product. Leftover frass can be utilized as a horticultural fertilizer or amendment during crop production. Crop growers sell or donate their products to other food partners, such as restaurants and grocery stores.
This study indicates that there is no impact on ornamental production or chemistry in a marigold color mix at a 10% BSFL frass incorporation. Therefore, without any treatment prior to incorporation, frass can replace 10% of a peat-based substrate without negatively impacting quality factors such as flower yield and antioxidants, which are the parameters of most interest to ornamental and medicinal producers. Additionally, plant size, weight, and greenness are not affected at a 10% incorporation rate. Future studies interested in evaluating commercially relevant cultivar mixes should run a color spectrum analysis to numerically quantify flower-color differences. Such information can then be used as a covariate whenever there is reason to suspect that flower coloration reflects physiological or experimental-unit differences. This creates opportunities to incorporate frass into commercial CEA production and to further increase research efforts to fully explore the impact of industry side streams on ornamental crops. Furthermore, frass amendments may help reduce the use of peat in an industry that relies on it heavily, but future research is needed to examine treatment methods for reducing salinity and ammonium stress.
The dataset of the study is available from the authors upon reasonable request.
Conceptualization, MYC; methodology, MYC, JH; formal analysis, MYC, CV-C; investigation, MYC, JH; resources, JH, JKC, JB; data curation, MYC; writing—original draft preparation, MYC; writing—review and editing, MYC, JH, JKC, JB, CV-C; visualization, MYC; project administration, JKC, JB; funding acquisition, JKC, JB. All authors have read and agreed to the published version of the manuscript.
The authors declare that there are no conflicts of interest.
This research was funded by Colorado State University Spur.
Great thanks to Rebekah Buena for her assistance in data collection and watering. Thank you to CSU Spur Soil, Water, and Plant Testing Lab in Denver, CO for completing our frass analysis.
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
12.
13.
14.
15.
16.
17.
18.
19.
20.
21.
22.
23.
24.
25.
26.
27.
28.
29.
30.
31.
32.
33.
34.
35.
36.
37.
38.
39.
40.
41.
42.
43.
44.
45.
46.
47.
48.
49.
50.
51.
52.
53.
54.
55.
56.
57.
58.
59.
60.
61.
62.
63.
64.
65.
66.
Chavez MY, Holley J, Craver JK, Velasco-Cruz C, Bousselot J. Increasing Rates of Black Soldier Fly (Hermetia illucens) Larval Frass Incorporation in African Marigold (Tagetes erecta) Production. J Sustain Res. 2026;8(1):e260041. https://doi.org/10.20900/jsr20260041.

Copyright © Hapres Co., Ltd. Privacy Policy | Terms and Conditions